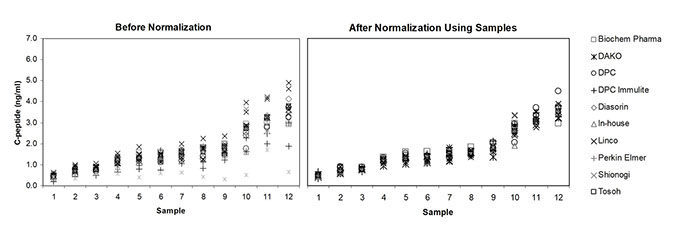
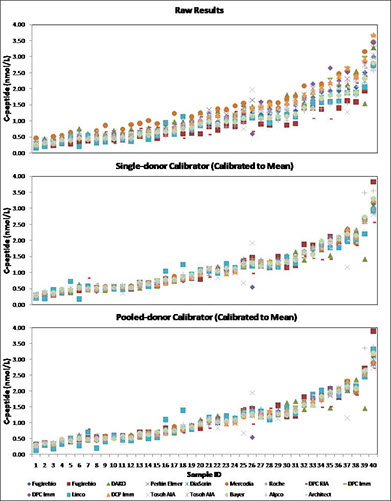
A Reference method was evaluated (x). Values were assigned to each sample (pooled and single-donor) and these were used as calibrators. Offline calibration with either single or pooled samples was shown to be effective in reducing variability of results from different methods (x).

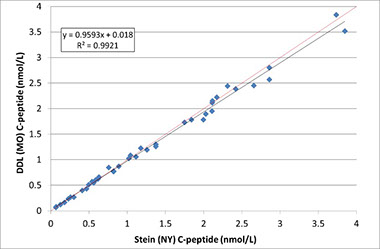
The Reference method described by Rogatsky, et al was established in a second laboratory, results from the two laboratories (DDL and Stein – Albert Einstein College of Medicine) were compared and this data was submitted to the JCTLM to be included in the list of Reference Measurement Procedures.
The JCTLM later included a Reference material submitted by the National Metrology Institute of Japan (NMIJ) (x). This material, CRM6901b, was then used to verify the results obtained by the DDL and the laboratory of Stein, et al (NY). Serum with zero C-peptide was spiked with CRM6901b and analyzed by LC/MS as a routine sample.
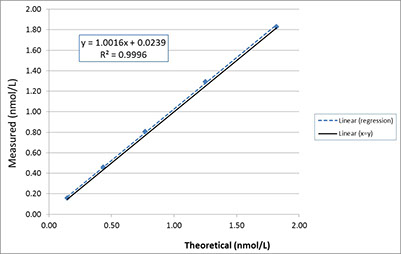
The NMIJ now also has a Reference Measurement Procedure on the JCTLM list. The DDL and NMIJ methods (both now traceable to CRM6901b) were compared.

References:
• Little RR, Kinumi T, Connolly SM, Kabytaev K. Implementing a Reference Measurement System for C-peptide: an Addendum. Clin Chem 2017: 63:1904-5.
• Little RR, Wielgosz RI, Josephs R, Kinumi T, Takatsu A, Li H, Stein D, Burns C. Implementing a Reference Measurement System for C-peptide: Successes and Lessons Learned. Clin Chem 2017;63:1447-56.
• Kabytaev K, Durairaj A, Shin D, Rohlfing CL, Connolly S, Little RR, Stoyanov AV. Two-step Ion-exchange chromatographic purification combined with reversed-phase chromatography to isolate C-peptide for mass Spectrometric analysis. J. Sep. Sci. 2016;39: 1-6.
• Stoyanov AV, Connolly S, Rohlfing CL, Rogatsky E, Stein D, Little RR. Human C-peptide Quantitation by LC-MS Isotope-Dilution Assay in Serum or Urine Samples. J Chromat Separation Techniq 2013;4:1-4.
• Stoyanov AV, Rohlfing CL, Connolly S, Roberts ML, Nauser CL, Little RR. Use of cation exchange chromatography for human C-peptide isotope dilution – Mass spectrometric assay. J Chromatogr. 1218;9244-9, 2011.
• Wiedmeyer HM, Polonsky KS, Myers GL, Little RR, Greenbaum CJ, Goldstein DE, Palmer JP. International Comparison of C-Peptide Measurements. Clin Chem, 53:784-787, 2007
• Bonser AM, Garcia-Webb P. C-peptide measurement and its clinical usefulness: a review. Ann Clin Biochem 1981;18:200-6.
• Palmer JP, Fleming GA, Greenbaum CJ, Herold KC, Jansa LD, Kolb H, et al. C-Peptide is the appropriate outcome measure for type 1 diabetes clinical trials to preserve -cell function: report of an ADA workshop, 21-22 October 2001. Diabetes 2004;53:250-64.
• Greenbaum CJ, Mandrup-poulsen T, McGee PF, Battelino T, Haastert B, Ludvigsson J, et al. Mixed-meal tolerance test versus glucagon stimulation test for the assessment of -cell function in therapeutic trials in type 1 diabetes. Diab Care 2008;31:1966-71.
• Lachin JM, McGee P, Palmer JP for the DCCT/EDIC Research Group. Impact of C-peptide preservation on metabolic and clinical outcomes in the Diabetes Control and Complications Trial. Diabetes 2014;63:739-48.
• Marx N, Silbernagel G, Brandenburg V, Burgmaier M, Kleber ME, Grammer TB, et al. C-peptide levels are associated with mortality and cardiovascular mortality in patients undergoing angiography: the LURIC study. Diab Care. 2013;36:708-14.
• Miller WG, Tate JR, Barth JH, Jones GRD. Harmonization: the sample, the measurement, and the report. Ann Lab Med 2014;34:187-97.
• Greenberg N. Update on current concepts and meanings in laboratory medicine – standardization, traceability and harmonization. Cln Chim Acta 2014;432:49-54.
• Bureau International des Poids et Mesures, Joint Committee for Traceability in Laboratory Medicine (JCTLM). http://www.bipm.org/en/committees/jc/jctlm/ (Accessed November 2016).
• Wiedmeyer HM, Polonsky KS, Myers GL, Little RR, Greenbaum CJ, Goldstein DE, Palmer JP. International comparison of C-peptide measurements. Clin Chem 2007;53:784-7.
• Little RR, Rohlfing CL, Tennill AL, Madsen RW, Polonsky KS, Myers GL, et al. Standardization of C-peptide measurements. Clin Chem 2008;54:1023-6.
• Rogatsky E, Balent B, Goswami G, Tomuta V, Jayatillake H, Cruikshank G, et al. Sensitive quantitative analysis of C-peptide in human plasma by 2-dimensional liquid chromatography – mass spectrometry isotope-dilution assay. Clin Chem 2006;52:872-9.
• Kinumi T, Ichikawa R, Hirokazu A,Takatsu A. Traceable amino acid analyses of proteins and peptides by isotope-dilution mass spectrometry using pre-column derivatization reagent. Anal Sci 2010;26:1007-10.
• Kato M, Yamazaki T, Kato H, Eyama S, Goto M, Yoshioka M, Takatsu A. Development of high-purity certified reference materials for 17 proteinogenic amino acids by traceable titration methods. Anal Sci 2015;31:805-14.
• Kinumi T, Goto M, Eyama S, Kato M, Kasamam T, Takatsu A. Development of SI-traceable C-peptide certified reference material NMIJ CRM 6901-a using isotope-dilution mass spectrometry-based amino acid analyses. Anal Bioanal Chem 2012;404:13-21.
• Kinumi T, Mizuno R, Takatsu A. Quantification of serum C-peptide by isotope-dilution liquid chromatography-tandem mass spectrometry: enhanced detection using chemical modification and immunoaffinity purification. J Chromatogr B Analyt Technol Biomed Life Sci 2014;953-4:138-42.
• Kuanysh, K., Maddaloni, E., Ferm, M.L., Redondo, M.J., Buzzetti, R., Pozzilli, P. C-peptide as an important biomarker in diabetes management: The need for standardization of its measurement. Diabetes Obes Metab 2026. DOI: 10.1111/dom.70447
• Rohlfing, C., Petroski, G., Hatten-Beck, M., Hanson, S., Hoofnagle, A.N., Little, R.R., Kuanysh, K. The current status of serum insulin measurements and the need for standardization. Clin Chem Lab Med 2025; 63(12):2442 – 2446. DOI: 10.1515/cclm-20250552
• Rohlfing, C., Petroski, G., Connolly, S.M., Hanson, S., Little, R.R., Kuanysh, K. A comparative analysis of current C-peptide assays compared to a reference method: can we overcome inertia to standardization? Clin Chem Lab Med 2025; 63(6):1124-1131. DOI: 10.1515/cclm-2024-1260
• Grothoff, W., Khodakivskyi, I., Shin, A., Little, R.R., Connolly, S.M., Kuanysh, K. MRM-based LC-MS method for accurate C-peptide quantitation. JMSACL 2025; 36: 1-8. DOI: 10.1016/j.jmsacl.2025.02.001
• Schleicher, E., Kuanysh, K., Nauck, M., Müller-Wieland, D., Peter, A., Little, R.R., Heinemann, L. Call for Standardization of C-Peptide Measurement. J Diabetes Sci Technol 2025; Aug 8:19322968251362848. doi: 10.1177/19322968251362848
• Moradian, A., Goonatilleke, E., Lin, T.-T., Hatten-Beck, M., Emrick, M., Schepmoes, A.A., Fillmore, T.L., MacCoss, M.J., Sechi, S., Sobhani, K., Little, R.R., Kuanysh, K., van Eyk, J.E., Qian, W.-J., Hoofnagle, A.N. Interlaboratory Comparison of Antibody-Free LC-MS/MS Measurements of C-peptide and Insulin. Clin Chem 2024; 70(6): 855 – 864. DOI: 10.1093/clinchem/hvae034
• Wu, Z., Kuanysh, K., Mu, J., Connolly, S.M., Clarke, N.J., Little, R.R., McPhaul, M.J. Critical need to assess modified and un-modified peptides in C-peptide standard materials. JMSACL 2022; 26:7 – 8. DOI: 10.1016/j.jmsacl.2022.07.003
• Little, R.R., Kuanysh, K., Connolly, S.M. Comments on Deng, et al: The potential for Isotope dilution-LC-MS/MS to improve laboratory measurement of c-peptide; reasons and critical determinants. JMSACL 2021; 22:79-80. DOI: 10.1016/j.jmsacl.2021.11.002
The NGSP is supported in part by National Institutes of Diabetes and Digestive and Kidney Diseases 1UC4DK096587-01.